Quick Summary
660nm LLLT could be a possible future Alzheimer’s treatment.
Device I use: 660nm Tendlite

Recent Study
A recent study by the world’s leading LLLT expert, Dr. Michael Hamblin, indicates that 660nm wavelength light activates white blood cells known as neutrophils and increases their ability to fight fungal infections (full abstract below).
Here’s what a wavelength is.
Neutrophils
Neutrophils are the most common white blood cells in mammals and are a part of the innate immune system.
Here’s what a neutrophil looks like.

The photoactivation of neutrophils increases their ability to kill invading fungi (candida albicans, for example).
Here’s a picture of that bugger.
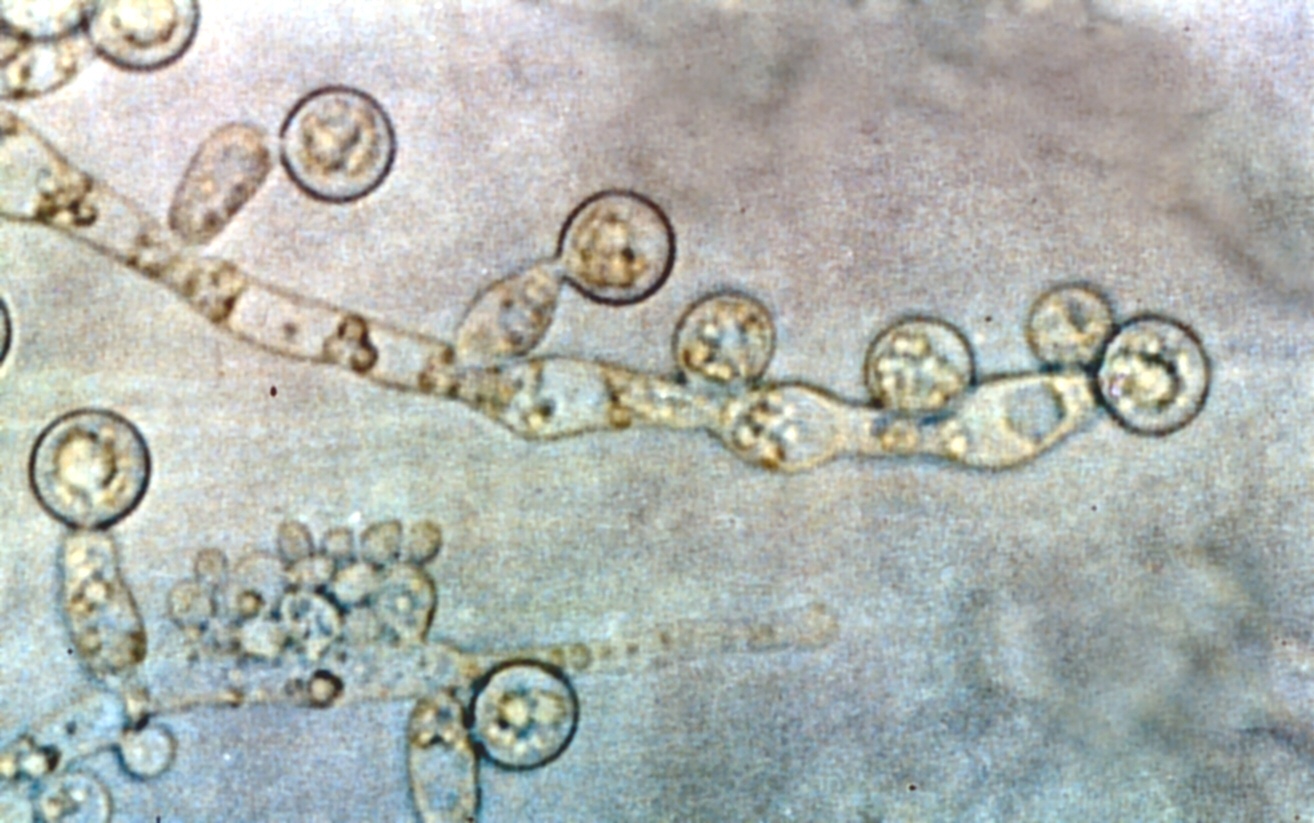
Fungal Theory of Alzheimer’s
Microbial infections (fungus, especially) have recently been correlated (causation is not fully established) with Alzheimer’s disease (abstracts below).
Could LLLT be used to enhance the body’s defense against fungus and therefore Alzheimer’s?
Possibly.
Definitely worth keeping eye on as more data comes out!
How I Use
I use the Tendlite on my forehead as well as on my carotid artery to photoexcite blood platelets (more about this in future posts) and white blood cells.

The Science
https://www.ncbi.nlm.nih.gov/pubmed/27243910
J Biophotonics. 2016 May 31. doi: 10.1002/jbio.201600035. [Epub ahead of print]
Low-level laser therapy stimulates the oxidative burst in human neutrophils and increases their fungicidal capacity.
Cerdeira CD1, Lima Brigagão MR1, Carli ML2, de Souza Ferreira C1, de Oliveira Isac Moraes G1, Hadad H2, Costa Hanemann JA2, Hamblin MR3,4,5, Sperandio FF6,7,8.
Author information
Abstract
Low-level laser therapy (LLLT) is known to enhance mitochondrial electron transfer and ATP production; thus, this study asked whether LLLT could stimulate the oxidative burst in human neutrophils (PMN) and improve their ability to kill microorganisms. Blood from healthy human subjects was collected and PMN were isolated from the samples. PMN were treated in vitro with 660 nm or 780 nm CW laser light at 40 mW power and increasing energies up to 19.2 J and were subsequently incubated with Candida albicans cells. Generation of hydroxyl radicals, hypochlorite anions and superoxide anions by PMN were checked using fluorescent probes and chemiluminescence assays; a microbicidal activity assay against C. albicans was also performed. LLLT excited PMN to a higher functional profile, which was translated as superior production of reactive oxygen species (ROS) and increased fungicidal capacity. The most efficacious energy was 19.2 J and, interestingly, the 660 nm light was even more efficacious than 780 nm at increasing the respiratory burst of PMN and the fungicidal capacity. Human neutrophils (PMN) were stimulated in vitro with 660 nm or 780 nm CW laser light at 40 mW of power and a total energy of 19.2 J. Low-level laser therapy (LLLT) excited PMN to a higher functional profile, which was translated as a superior production of reactive oxygen species (ROS) such as hydroxyl radicals (HO• ) and hypochlorite anions (ClO- ) (Figure) and increased fungicidal capacity against Candida albicans cells.
http://stm.sciencemag.org/content/8/340/340ra72
Sci Transl Med. 2016 May 25;8(340):340ra72. doi: 10.1126/scitranslmed.aaf1059.
Amyloid-β peptide protects against microbial infection in mouse and worm models of Alzheimer’s disease.
Kumar DK1, Choi SH1, Washicosky KJ1, Eimer WA1, Tucker S1, Ghofrani J1, Lefkowitz A1, McColl G2, Goldstein LE3, Tanzi RE4, Moir RD4.
Author information
Abstract
The amyloid-β peptide (Aβ) is a key protein in Alzheimer’s disease (AD) pathology. We previously reported in vitro evidence suggesting that Aβ is an antimicrobial peptide. We present in vivo data showing that Aβ expression protects against fungal and bacterial infections in mouse, nematode, and cell culture models of AD. We show that Aβ oligomerization, a behavior traditionally viewed as intrinsically pathological, may be necessary for the antimicrobial activities of the peptide. Collectively, our data are consistent with a model in which soluble Aβ oligomers first bind to microbial cell wall carbohydrates via a heparin-binding domain. Developing protofibrils inhibited pathogen adhesion to host cells. Propagating β-amyloid fibrils mediate agglutination and eventual entrapment of unatttached microbes. Consistent with our model, Salmonella Typhimurium bacterial infection of the brains of transgenic 5XFAD mice resulted in rapid seeding and accelerated β-amyloid deposition, which closely colocalized with the invading bacteria. Our findings raise the intriguing possibility that β-amyloid may play a protective role in innate immunity and infectious or sterile inflammatory stimuli may drive amyloidosis. These data suggest a dual protective/damaging role for Aβ, as has been described for other antimicrobial peptides.
https://www.ncbi.nlm.nih.gov/pubmed/26468932
Sci Rep. 2015 Oct 15;5:15015. doi: 10.1038/srep15015.
Different Brain Regions are Infected with Fungi in Alzheimer’s Disease.
Pisa D1, Alonso R1, Rábano A2, Rodal I2, Carrasco L1.
Author information
Abstract
The possibility that Alzheimer’s disease (AD) has a microbial aetiology has been proposed by several researchers. Here, we provide evidence that tissue from the central nervous system (CNS) of AD patients contain fungal cells and hyphae. Fungal material can be detected both intra- and extracellularly using specific antibodies against several fungi. Different brain regions including external frontal cortex, cerebellar hemisphere, entorhinal cortex/hippocampus and choroid plexus contain fungal material, which is absent in brain tissue from control individuals. Analysis of brain sections from ten additional AD patients reveals that all are infected with fungi. Fungal infection is also observed in blood vessels, which may explain the vascular pathology frequently detected in AD patients. Sequencing of fungal DNA extracted from frozen CNS samples identifies several fungal species. Collectively, our findings provide compelling evidence for the existence of fungal infection in the CNS from AD patients, but not in control individuals.
https://www.ncbi.nlm.nih.gov/pubmed/25125470
J Alzheimers Dis. 2015;43(2):613-24. doi: 10.3233/JAD-141386.
Direct visualization of fungal infection in brains from patients with Alzheimer’s disease.
Pisa D1, Alonso R1, Juarranz A2, Rábano A3, Carrasco L1.
Author information
Abstract
Recently, we have reported the presence of fungal infections in patients with Alzheimer’s disease (AD). Accordingly, fungal proteins and DNA were found in brain samples, demonstrating the existence of infection in the central nervous system. In the present work, we raised antibodies to specific fungal species and performed immunohistochemistry to directly visualize fungal components inside neurons from AD patients. Mice infected with Candida glabrata were initially used to assess whether yeast can be internalized in mammalian tissues. Using polyclonal rabbit antibodies against C. glabrata, rounded immunopositive cells could be detected in the cytoplasm of cells from liver, spleen, and brain samples in infected, but not uninfected, mice. Immunohistochemical analyses of tissue from the frontal cortex of AD patients revealed the presence of fungal material in a small percentage (~10%) of cells, suggesting the presence of infection. Importantly, this immunopositive material was absent in control samples. Confocal microscopy indicated that this fungal material had an intracellular localization. The specific morphology of this material varied between patients; in some instances, disseminated material was localized to the cytoplasm, whereas small punctate bodies were detected in other patients. Interestingly, fungal material could be revealed using different anti-fungal antibodies, suggesting multiple infections. In summary, fungal infection can only be observed using specific anti-fungal antibodies and only a small percentage of cells contain fungi. Our findings provide an explanation for the hitherto elusive detection of fungi in AD brains, and are consistent with the idea that fungal cells are internalized inside neurons.
Where to Buy



2 Comments
Leave your reply.